CBSE Test Paper 01
Chapter 11 Thermal Properties of Matter
-
Molar Specific Heat capacity has units of 1
- W/m.K
- J/mol.K
- J.ohm/sec.K2
- J/kg.K
-
A hole is drilled in a copper sheet. The diameter of the hole is 4.24 cm at 27.0oC. What is the change in the diameter of the hole when the sheet is heated to 227oC? Coefficient of linear expansion of copper = 1.70 10-5 K–1. 1
- 1.24 x 10-2 cm
- 1.44 x 10-2 cm.
- 1.34 x 10-2 cm
- 1.54 x 10-2 cm
-
A student eats a dinner rated at 2000 Calories. He wishes to do an equivalent amount of work in the gymnasium by lifting a 50.0-kg barbell. How many times must he raise the barbell to expend this much energy? Assume that he raises the barbell 2.00 m each time he lifts it and that he regains no energy when he drops the barbell to the floor. 1
- 8.14 103
- 8.74 103
- 8.54 103
- 8.34 103
-
A spray can containing a propellant gas at twice atmospheric pressure (202 kPa) and having a volume of 125 cm3 is at 22oC. It is then tossed into an open fire. When the temperature of the gas in the can reaches 195oC, what is the pressure inside the can? Assume any change in the volume of the can is negligible. 1
- 260 kPa
- 300 kPa
- 290 kPa
- 320 kPa
-
A steel railroad track has a length of 30.000 m when the temperature is 0.0oC. Suppose that the ends of the rail are rigidly clamped at 0.0oC so that expansion is prevented. What is the thermal stress set up in the rail if its temperature is raised to 40.0oC? = 11 10-6 (C)-1 Y-steel = 20GPa 1
- 8.4 106 N/m2
- 8.1 107 N/m2
- 9.1 107 N/m2
- 8.8 106 N/m2
- Why the temperature above 1200°C cannot be measured accurately by a platinum resistance thermometer? 1
- Stainless steel cooking pans are preferred with extra copper bottom. Why? 1
- Each side of a cube increases by 0.01% on heating. How much is the area of its faces and volume increased? 1
- Calculate the heat of combustion of coil when 10 g of coal on burning raises the temperature of 2 litres of water from 20o C to 55o C. 2
- An iron ring of diameter 5.231 m is to be fixed on a wooden rim of diameter 5.243 m both initially at 27°C. To what temperature should the iron ring be heated so as to fit the rim? (Coefficient of linear expansion of iron is 1.2 10-5 K-1) 2
- What do you mean by molar specific heat of a substance? What is its SI unit? 2
- A ‘thermacole’ icebox is a cheap and efficient method for storing small quantities of cooked food in summer in particular. A cubical icebox of side 30 cm has a thickness of 5.0 cm. If 4.0 kg of ice is put in the box, estimate the amount of ice remaining after 6 h. The outside temperature is 45 °C, and co-efficient of thermal conductivity of thermacole is 0.01 J s-1m-1K-1. [Latent heat of melting of ice = 335 103 Jkg-1] 3
- A ‘thermacole’ icebox is a cheap and efficient method for storing small quantities of cooked food in summer in particular. A cubical icebox of side 30cm has a thickness of 5.0 cm. If 4.0kg of ice is put in the box, estimate the amount of ice remaining after 6h. The outside temperature is 45°C, and co-efficient of thermal conductivity of thermacole is 0.01 J s-1 m-1K-1. [Heat of fusion of water = 335 103 J kg-1] 3
- What is calorimetry? Briefly explain its principle. 3
- A rail track made of steel having length 10 m is clamped on a railway line at its two ends (Figure).
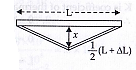
On a summer day due to rise in temperature by 20° C, it is deformed as shown in figure. Find x (displacement of the centre) if = 1.2 10-5 /℃. 5
CBSE Test Paper 01
Chapter 11 Thermal Properties of Matter
Answer
-
- J/mol.K
Explanation:
- J/mol.K
-
- 1.44 x cm.
Explanation: Change in diameter
= 1.70 10-5 4.24 (227-27) = 1.44 10-2 cm
- 1.44 x cm.
-
- 8.54 103 times
Explanation: Total no.of times he lift barbell = Total energy/energy in lifting barbell once
= 4.2 103 times
- 8.54 103 times
-
- 320 kPa
Explanation:
= 320KPa
- 320 kPa
-
- 8.8
Explanation: Thermal Stress
= 20 109 11 10-6 40 = 8.8 106
- 8.8
-
The temperature above 1200°C cannot be measured accurately by a platinum resistance thermometer because platinum begins to evaporate above 1200°C.
-
The copper bottom allows more heat to flow into the pan because the thermal conductivity of copper is much larger than that of steel. Therefore, it helps in cooking the food faster.
-
As each side of a cube increases by 0.01% on heating. This results in increased in the area of faces by 0.02% and the volume by 0.03%.
-
As per question mass m of 2 litres of water = 2 kg and we know that specific heat of water
The heat energy required to raise the temperature of water from 20o C to 55o C
= 2 4200 (55 – 20) = 2.94 105 j
As this much heat is produced by combustion of mo = 10g of coal, hence heat of combustion of
coal = -
From the definition of coefficient of linear expansion,
5.243 = 5.231[1 + 1.2 10-5 (T – 300)]
T = 191 + 300 = 491 K = 218°C -
Molar Specific heat
It is defined as the amount of heat energy required to raise the temperature of of that substance through .
Molar specific heat
where = the amount of heat energy required to raise the temperature of per moles of the given
substance through .
Its SI unit is -
Side of the given cubical ice box, s = 30 cm = 0.3 m
Thickness of the ice box, = 5.0 cm = 0.05 m
Mass of ice kept in the ice box, m = 4 kg
Time gap, t = 6 h
Outside temperature, T1= 45oC
Inside temperature, T2 = temperature of ice = 0oC
Coefficient of thermal conductivity of thermacole, K = 0.01 J s-1m-1K-1
Latent heat of melting of ice,
Let m’ be the total amount of ice that melts in 6 h.
Total amount of heat lost, in 6 hrs by the food(mathematical form of the equation comes from the defination of the thermal conductivity):
Where, A= Total surface area of the box = 6 × Surface area of each surface of the box=
But , from the defination of latent heat of melting of ice.
Mass of ice left = Mass of ice initially kept inside the box – Mass of ice melted in 6hrs = 4 – 0.313 = 3.687 kg
Hence, the amount of ice remaining after 6 h is 3.687 kg. -
Side of the given cubical ice box, s = 30 cm = 0.3m
Area of each surface of the box = s2 = (0.3)2 m3
Thickness of the icebox, = 5.0 cm = 0.05 m
Mass of ice kept in the icebox, m = 4kg
Time gap, t = 6 h = 6 60 60s
Outside temperature, = 45°C
Inside temperature, = 0℃
Coefficient of thermal conductivity of thermacole, K = 0.01 J s-1 m-1K-1
Heat of fusion of water, L = J kg-1
Let ‘m’ be the total amount of ice that melts in 6h.
The amount of heat lost by the food, kept in the thermacole in 6 hrs(using the equation from defination of thermal conductivity):
Where, A = Total surface area of the box = 6 × area of each surface = 6 (0.3)2 = 0.54 m3
Now, if m’ be the mass of melted ice, then
Mass of ice left = 4 – 0.313 = 3.687 kg
Hence, the amount of ice remaining after 6h is 3.687kg. -
Calorimetry is a branch of physics that deals with the measurement of the quantity of heat being transferred from one substance to another.
The principle of calorimetry follows the law of conservation of energy. According to the principle of calorimetry, if two chemically non-reactive substances maintained at different temperatures are brought in thermal contact so as to share heat, the hotter substance transfers heat to the colder substance and the heat exchange continues till the temperature of both the substances become same. In such a situation if there is no heat energy dissipated due to radiation loss etc., then, we have
Total heat transferred from the hotter body = Total heat transferred to the colder body. -
Coefficient of linear expansion initial length (change in temperature)
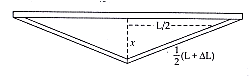
From the above figure using Pythagoras theorem, perpendicular2 = hypotenuse 2 – base2, we get
…..(i)
Again from the definition of coefficient of linear expansion, change in length due to increase in temperature,, since = initial length at 0℃ coefficient of linear expansion increase in temperature
Now putting the values of we get, m
m
,
This is the required displacement of the centre