CBSE Test Paper 01
Chapter 12 Thermodynamics
-
Consider the cyclic process depicted in Figure If Q is negative for the process BC, and if is negative for the process CA, what are the signs of W and that are associated with BC? 1
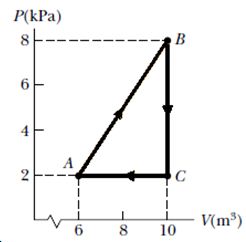
- 0, +
- 0, –
- +, –
- 0, –
-
One of the most efficient engines ever built (actual efficiency 42.0 percent) operates between 430oC and 1870oC. How much power does the engine deliver if it absorbs 1.40 J of energy each second from the hot reservoir? 1
- 52.8 kW
- 62.8 kW
- 58.1 kW
- 58.8 kW
-
A power plant that would make use of the temperature gradient in the ocean has been proposed. The system is to operate between 5.00oC (water temperature at a depth of about 1 km) and 20.0oC (surface water temperature).What is the maximum efficiency of such a system? 1
- 5.02 percent
- 5.12 percent
- 5.45 percent
- 5.32 percent
-
A 2.00-mol sample of helium gas initially at 300 K and 0.400 atm is compressed isothermally to 1.20 atm. Assuming the behavior of helium to be that of an ideal gas, find the energy transferred by heat 1
- -5.38 kJ
- -5.28 kJ
- -5.58 kJ
- -5.48 kJ
-
Internal energy of a system is 1
- a complex variable
- a random variable
- a state variable
- a discrete variable
-
State Carnot’s Theorem? 1
-
State zeroth law of thermodynamics? 1
-
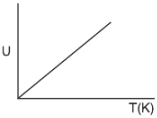
Plot a graph between internal energy U and Temperature (T) of an ideal gas. 1
-
What is thermodynamics? State its important characteristics. 2
-
Consider the cyclic process ABCA on a sample 2 mol of an ideal gas as shown. The temperature of the gas at A and B are 300K and 500K respectively. Total of 1200 J of heat is drawn from the sample. Find the work done by the gas in part BC. 2
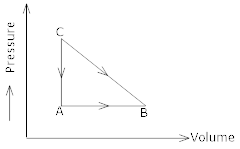
-
A gas is filled in a cylinder at 300 K. Calculate the temperature upto which it should be heated so that its volume becomes of its initial volume. 2
-
An indirectly heated filament is radiating maximum energy of wavelength 2.1610-7m. Find the net amount of heat energy lost per second per unit area, the temperature of surrounding air is 130C. Given b = 2.8810-3 mk, = 5.7710-8 J / sec/ m2 / k4 ? 3
-
If for hydrogen CP – CV = a and for oxygen CP – CV = b where CP & CV refer to specific heat at constant pressure and volume then what is the relation between a and b? 3
-
How do you derive Newton’s law of cooling from Stefan’s law? 3
-
Derive the equation of state for adiabatic change. 5
CBSE Test Paper 01
Chapter 12 Thermodynamics
Answer
-
-
0, –
Explanation: W = area under P-V curve = 0
Q = Eint + W
W = 0, Q = -ve
Eint = -ve
-
-
-
58.8 kW
Explanation:
W = 58.8 KJ
Power = Energy (or work) transfer per second = 58.8KW
-
-
-
5.12 percent
Explanation:
-
-
-
-5.48 kJ
Explanation:
in isothermal process
= – 5.48 KJ
-
-
-
a state variable
Explanation: A state function describes the equilibrium state of a system. For example, internal energy, enthalpy, and entropy are state quantities because they describe quantitatively an equilibrium state of a thermodynamic system, irrespective of how the system arrived in that state.
-
-
According to Carnot’s Theorem, no engine working between two temperatures can be more efficient than a Carnot’s reversible engine working between the same temperatures.
-
According to this, when the thermodynamic system A and B are separately in thermal equilibrium with a third thermodynamic system C, then the system A and B are in thermal equilibrium with each other as well.
-
-
Thermodynamics is the branch of physics that deals with the concepts of heat and temperature and the interconversion of heat into other forms of energy. Important characteristics (features) of thermodynamics are as follows :
- It is a macroscopic science and deals with the bulk system.
- Thermodynamics does not go into the molecular constitution of matter.
- The state of a thermodynamic system is specified by macroscopic variables like pressure, volume, temperature, mass etc., that is felt by our sense perceptions and are measurable.
-
The change in internal energy during the cyclic process is zero. Therefore, heat supplied to the gas is equal to work done by it,
WAB + WBC + WCA = – 1200J …(i)
(- ve because the cyclic process is traced anticlockwise the net work done by the system is negative)
The work done during the process AB is
WAB = PA (VB-VA) = nR(TB-TA) [QPV = nRT]
WAB = 2 8.3(500-300) = 3320J ….(ii)
R = Universal gas constant
N = No. of volume
Since in this process, the volume increases, the work done by the gas is positive.
Now, WCA = O ( volume of gas remains constant)
3320 + WBC + O = – 1200 (Using equation (i) & (ii)
WBC = – 1200 – 3320
WBC = – 4520J -
Given,,
,
According to Charles Law, -
From Wien’s Law:-
T = constant = b ……….. (i)
b – Wien’s constant
b = 2.910-3 m K
Now,
Temperature of filament = T
So, [by equation (i)]
= 13333.3K
Now surrounding temperature = To = 13 + 273 = 286 K.
Let total amount of heat energy lost per second per unit area = E
By Stefan’s law:
= 1.824 108 W/m2
Hence the energy lost per second per unit area of the body is 1.824 108 W/m2 -
Let
CP = Specific heat at constant pressure
CV = Specific heat at constant Volume
M = Molar mass
For an ideal gas , Cp – Cv = R
for other gases,
For H2, CP – CV = a
For O2 = CP – CV = b
So, for
…(i)
For …(ii)
from equation (i)
from equation (ii)
2a = 32 b
a = 16 b -
Acc. to Newton’s law of cooling, the time rate of loss of heat of a liquid is directly proportional to the difference in temperature of the liquid and the surrounding, provided the difference in temperature is very small.
Let a body be maintained at T K. Let To be the temperature of the surroundings. Let T ≫ To. There will be a loss of heat by the body
Acc. to Stefan’s law, amount of heat energy lost per second per unit area of the body is
(T4 – T04)
= Stefan’s constant
= Emissivity of the body and surroundings
[ (a4 – b4) = (a2 – b2)(a2 + b2) ]
(T2 – T02)(T2 + T02)
In case of Newton’s cooling, when temperature difference between body and its surrounding is not very large T To
(T – T0)(T + T0)(T02 + T02)
E = K(T – To)
Hence the Newton’s law of cooling -
Consider 1 mol of an ideal gas enclosed in a cylinder fitted with a perfectly frictionless piston.
Let P = initial pressure, V = initial volume and T = Initial temperature of the gas.
Suppose a small amount of heat dQ is given to the system. The heat is spent in two ways:-- The heat required in increasing temperature of the gas in very small range dT at constant volume = CvdT
- The heat required in the expansion of the gas by very small volume dV = PdV
So, dQ = CV dT + PdV
In an adiabatic process, no heat exchange takes place,
So, dQ = 0
CvdT + PdV = 0 …(i)
According to the standard gas equation,
PV = RT
Differentiate both sides,
PdV + VdP = RdT + TdR
(dR = 0, as R is a gas constant)
Using this equation in (i)
CvPdV + CvVdP + RPdV = 0
(Cv + R) PdV + CvVdP = 0 …(ii)
As, Cp – Cv = R
or Cp = R + Cv
So equation (ii) becomes
CpPdV + CvVdP = 0
Divide above equation by CvPV
Now, integrate both side,
Loge V + Loge P = Constant
Loge + LogeP = Constant
( aLogeb = Logeba)
Loge = Constant
( Logea + Logeb = Logeab)
= e(Constant)
where, k = another constant